Photo-Excited Toluidine Blue Disaggregates the Repeat Tau and Modulates End-Binding Protein EB1, Cytoskeletal Structure in Neuronal Cells
bThe School of Bioengineering Sciences and Research, MIT Art, Design & Technology, University, Pune, India,
cAcademy of Scientific and Innovative Research (AcSIR), Ghaziabad, India,
dDepartment of Neurochemistry, National Institute of Mental Health and Neuro Sciences (NIMHANS), Institute of National Importance, Bangalore, Karnataka, India
Keywords
Abstract
Background/Aims:
Alzheimer’s disease is a progressive neurological disorder characterized by the intracellular accumulation of Tau protein aggregates. In the present work, we studied the effect of Toluidine Blue and photo-excited Toluidine Blue on the aggregation of repeat Tau using in vitro assays.Methods:
The in vitro experiments were carried out on recombinant repeat Tau which was purified by cation exchange chromatography. The ThS fluorescence analysis was used to study the aggregation kinetics of Tau. CD spectroscopy and electron microscopy were used to study the secondary structure and morphology of Tau respectively. The actin cytoskeleton modulation was studied in Neuro2a cells with help of immunofluorescent microscopy.Results:
Results showed that Toluidine Blue efficiently inhibited the formation of higher-order aggregates, which was evidenced by Thioflavin S fluorescence assay, SDS-PAGE, and TEM. Immunofluorescence studies on the cytoskeleton of Neuro2a cells showed that Toluidine Blue and photo-excited Toluidine Blue treatment at a non-toxic concentration of 0.5 µM stimulated the formation of actin-rich lamellipodia and filopodia structures. Tubulin networks were also differentially modulated after the treatment of Toluidine Blue and photo-excited Toluidine Blue. End-binding protein 1 (EB1) levels were observed to increase after Toluidine Blue and photo-excited Toluidine Blue treatment indicating accelerated microtubule polymerization.Conclusion:
The overall study suggested that Toluidine Blue inhibited the aggregation of soluble Tau and photo-excited Toluidine Blue disaggregated the pre-formed Tau filaments. In our study, TB and PE-TB were observed to be potent against Tau aggregation. We observed a distinctive modulation of actin, tubulin networks, and EB1 levels after TB and PE-TB treatment, which suggested that TB and PE-TB have potency against cytoskeleton deformities.Introduction
Alzheimer’s disease (AD) is characterized by the accumulation of neurofibrillary tangles (NFTs), which are aggregates of intracellular Tau [1, 2]. The aggregated Tau has been reported to be involved in several other neurodegenerative diseases including pick’s disease, corticobasal degeneration, progressive supranuclear palsy, post-encephalitic parkinsonism, etc. , which are termed Tauopathies [3, 4]. Tau is a natively unfolded cytoskeleton-associated protein having the physiological role of stabilizing microtubules and cytoskeleton structures [5]. Neurodegenerative diseases have been reported to be closely associated with cytoskeleton abnormalities in neuronal cells involving actin and tubulin deformities [6, 7]. Actin and tubulin are abundant cytoskeleton proteins. (Bray & Gilbert 1981). Tubulin is known to involve in the formation of microtubules whereas, actin majorly assists the cells in substratum adhesion, synapse formation, and cel,l motility by the formation of structures such as lamellipodia, filopodia, podosomes etc [8, 9]. End-binding protein 1 (EB1) is another class of cytoskeletal-associated protein, which has been reported to be located at the growing end of microtubules [10]. Among all these cytoskeleton proteins microtubule-associated protein Tau and its aggregates are majorly focused in the context of the generation of neuronal abnormalities. Tau protein has a domain organization comprising of a projection domain and microtubule-binding domain. Microtubule-binding domain of Tau has 4 repeat region, which is considered as the aggregation prone region of Tau (Gustke et al. 1994). The hexapeptide VQIINK and VQIVYK present in the 2nd and 3rd repeat of Tau respectively, are reported to be involved in Tau aggregation [11, 12]. To study the aggregation inhibition propensity of certain molecules against Tau, the repeat region of Tau has been targeted [13, 14]. Several compounds of natural and synthetic origin have been studied against Tau aggregation [15, 16]. These compounds can be categorized in two groups based on their mode of action, firstly disaggregation of the mature Tau fibrils and the other by inhibition of the aggregation of Tau as reported for anthraquinones and EGCG respectively [17]. Several dyes such as methylene blue, Toluidine Blue (TB), and rose bengal have been reported to modulate Amyloid-β peptides aggregation, which is the other hallmark protein of AD [17-19]. Phthalocyanine dye has been reported to be effective against pathological prion protein (PrPC) [20]. Moreover, photo-active chlorin 6 dye was also found to reduce the aggregation by modulating the histidine residues in which protein (Amyloid-β) [21]. Methylene blue derived Leuco-methylthioninium Bis (Hydromehanesulphonate) (LMTM) entered phase-3 clinical trial for the treatment of mild AD [22] However, LMTM in further studies was found to be ineffective [23]. Methylene blue and its derivatives are neuroprotective molecules [29, 30]. TB is a known basic dye used in histological staining but the medicinal properties of TB have not been studied illustratively. TB is a phenothiazine dye having structural similarity with methylene blue. The basic dye TB has been reported for its application in histology. TB has an affinity for nucleic acid and it majorly binds to tissues rich in DNA and RNA. TB has been widely applied for vital staining which facilitates the detection of early malignant lesions [24]. Several studies have revealed the anti-bacterial and anti-biofilm properties of TB but, the role of TB in neurodegeneration has not been explored extensively. The recently published literature has supported the in vitro aggregation inhibition and disaggregation potency of TB and PE-TB against full-length Tau. Moreover, the treatment of PE-TB also improved memory and behavioral deficits in the transgenic drosophila model [25]. In the present study, we evaluated the potency of TB against repeat Tau aggregation. Furthermore, the effect of TB in the presence of irradiation was also studied in disaggregating the matured fibrillary aggregates of repeat Tau. In the present study, the potency of TB and PE-TB against Tauopathy was studied in various aspects.
Materials and Methods
Materials
MES (M3671), BSA (82516), BES (14853), BCA (B9643), CuSO4 (C2284) ThS (T1892), Toluidine Blue (T3260), MTT (M2128) were purchased from Sigma. IPTG (420322) and DTT (3870) were purchased from Calbiochem. Other chemicals such as Ampicillin (2007081), NaCl (194848), KCl (194844), Na2HPO4 (191437), KH2PO4 (19142), EGTA (194823), MgCl2 (191421), PMSF (195381), Ammonium acetate (191404), Heparin (904108) and DMSO were purchased from MP Biomedicals and protease inhibitor cocktail was from Roche. Copper-coated carbon grids were purchased from Ted Pella (01814F, carbon type-B, 400 mesh, Cu). DMEM advanced F12 media (12634010), Fetal bovine serum (16000044), Pensterp cocktail (04693159001), and Anti-anti (15240062) were purchased from Gibco.
Purification of recombinant Repeat Tau
The recombinant Tau expressed in E.coli BL 21* cells was purified by the method suggested in earlier literature [15]. Briefly, the E.coli was grown in Luria-Bertani broth at 37°C, at 180 rpm in an orbital shaker (INFORS HT) till the culture obtained OD600 of 0.5. In the log phase, the culture was induced with 0.5 mM isopropyl β-D-1-thiogalactopyranoside (IPTG) and incubated for further 4 hours at 37 °C in an orbital shaker at 180 rpm. Further, 4 hours of post-induction incubation time was followed according to the standard protocol [17]. The culture was pelleted down by centrifuging at 4000 rpm for 10 minutes in Avanti JXN 26. The pellets were further suspended in lysis buffer (50 mM MES, 1 mM EGTA, 2 mM MgCl2, 5 mM DTT, 1 mM PMSF, and 50 mM NaCl), and the lysis was carried out under high pressure. The culture suspension was lysed at 15000 psi of pressure in a homogenizer (Constant cell disruption system) with a continuous lysis cycle. After 2 cycles of lysis, the lysate was collected and stored on ice for reducing the protein degradation. The subsequent step of lysis is the lysate heating at 90°C for 20 min in a water bath (Benchmark) without any agitation. After heating the lysate was allowed to cool and then centrifuged at 164, 700 × g (Optima XPM ultracentrifuge, Beckman Coulter) for 45 min at 4°C. The supernatant was collected and dialyzed against buffer A. Tau is a positively charged protein, hence the purification was done by cation-exchange chromatography as described in previous studies [35]. The repeat Tau was eluted by giving a linear gradient of 1 M NaCl, and the fraction under the elution peak was collected and concentrated. The desired molecular weight of our protein was 13.4 kDa, hence the quality of the protein was observed on 17% sodium dodecyl sulfate-polyacrylamide gel (SDS-PAGE). The concentration of protein was estimated by bicinchoninic acid assay (BCA assay) [36]. Briefly, bovine serum albumin (BSA) was diluted for making the standard graph. Tau protein was diluted in ratios of (1:100, 1:200, and 1:400). BCA reagent was prepared freshly by mixing bicinchoninic acid and CuSO4 in the ratio of 4:1. After the addition of BCA to protein, the mixture was incubated at 37°C for 60 minutes in dark. The absorbance was measured at 562 nm in Tecan M 200 multimode plate reader and concertation was estimated referring to the standard graph.
Preparation of repeat Tau-fibrils
The soluble repeat Tau was induced to form aggregates by incubating with heparin as suggested in the published literature [17, 37]. 100 µM of soluble repeat Tau was incubated with 25 µM heparin. The assembly was carried out in 20 mM BES buffer supplemented with 1 mM DTT, 25 mM NaCl, and a protease inhibitor cocktail. The assembly mixture was kept at 37°C for 3 days and the aggregates were confirmed by SDS-PAGE and electron microscopy. The heterogeneous Tau aggregates were observed on SDS-PAGE as bands of higher molecular weight (25-150 kDa) [38]. Moreover, the electron microscopy images suggested the presence of long-tangled Tau filaments, which supported the presence of aggregates in our sample.
ThS fluorescence assay
Thioflavin S (ThS) assay was used to monitor the assembly of Tau fibrils. The procedure was followed according to earlier published work [17]. The Tau protein was measured at a concentration of 2 µM incubated with 8 µM ThS dissolved in ammonium acetate. The mixture was incubated at room temperature in dark for 10 min and the fluorescence was measured at an excitation wavelength of 480 nm and emission was measured at 520 nm in Tecan M 200 multimode plate reader.
Electron microscopy
The morphological analysis of Tau fibrils was done by scanning under transmission electron microscopy. Tau was incubated at a concentration of 2 µM on 400 mesh copper-coated carbon grid for 90 seconds. After 2 subsequent washes with milliQ, the grid was incubated with 2% uranyl acetate for 120 seconds. The grids were blot-dried and kept at room temperature. The scanning was done using a Tecnai T20 electron microscope.
Photo-irradiation of TB
TB has an absorption maxima of 630 nm, thus it was photo-excited by a red light. For photo-excitation, a dark chamber was designed, which comprises a commercially procured red LED light source (3.5 watts). The setup was equipped with a thermometer to measure heat changes during the irradiation. The repeat Tau aggregates were mixed with various concentrations of TB (2-40 µM). The mixture was transferred to a 96-well black well plate. The plate was exposed to red light in the dark chamber for the photo-excitation of TB. After 180 minutes of irradiation, the samples were taken out from the plates and analyzed by various biochemical and biophysical assays including SDS-PAGE, ThS fluorescence assay, and electron microscopy. The irradiation dose or irradiance was calculated by the formula
Where E is irradiance, time was calculated in terms of seconds
Cell biology studies
Mouse neuroblastoma cells Neuro2a (ATCC: CCL-131), were cultured in advanced DMEM F12 media. 104 cells/well were seeded in 96 well plates for the viability assay. The cells were treated with various concentrations of TB viz, 1, 2.5, 5, 10, 20, 40, 80, and 120 µM for 24 hours. After the incubation, 10 µl of 5 mg/ml MTT solution was added to each well. The plate was further incubated at 37°C for 4 hours. The formazan crystals were dissolved in 100% DMSO and the absorbance was measured at 570 nm.
Immunofluorescence analysis
Neuro2a cells (50, 000/well) were seeded on a glass coverslip and allowed to incubate for 24 hours at 37°C. The cells were treated with various concentrations of TB (0.5 and 50 µM) and irradiated for 10 minutes with a red light. After the irradiation, cells were incubated further 24 hours and processed for immunostaining. The treated and untreated cells were fixed with absolute methanol for 20 minutes at -20°C. 0.2% Triton X-100 was used for cell permeabilization. For avoiding the non-specific binding of the antibody, the cells were incubated with 5% horse serum for 1 hour. The cells were incubated with anti-tubulin (Thermo PA1-41331; dilution 1:250) and K9JA (Dako A0024; dilution 1:500) antibodies. After overnight incubation, the cells were incubated secondary antibody tagged with Alexa Fluor 488 (Thermo, A11034) (anti-rabbit; dilution 1:1000) and Alexa Fluor 555 (Thermo, A32727) (Anti-mouse; dilution 1:500). The nucleus was stained with DAPI. After the incubation coverslips were mounted using mounting media (70% glycerol) and were sealed on a glass slide. These slides were allowed to air dry at room temperature before the scanning. These samples were scanned on Zeiss Axio observer inverted microscope using 63X magnification in oil emersion and at 40% light intensity.
Disaggregation of PE-TB
TB-treated repeat Tau aggregates were irradiated with red light for 180 minutes. The PE-TB treated aggregates and untreated samples were incubated with 8 µM of ThS dye (diluted in 50 mM ammonium acetate) for 10 minutes in dark. The samples were transferred in 384 black well plates and the fluorescence measurement was recorded on excitation of 480 and emission of 521 nm. For further analysis the PE-TB treated aggregates and untreated control aggregates were loaded on 10% SDS-PAGE. The SDS-PAGE was stained with 1% coomassie brilliant blue solution for 10 minutes, after destaining the gel was analyzed for the presence of higher-order aggregates in treated and untreated samples.
Study of the effect of PE-TB on the cytoskeleton
The effect of TB and PE-TB on the cytoskeleton was observed in Neuro2a cells. The cells were treated with 0.5 µM TB and were irradiated for 10 minutes. The treated cells were incubated at 37°C for 24 hours and the effect on cytoskeleton modulation was monitored by immunofluorescence and western blot analysis.
Statistical Analysis
The statistical data for the fluorescence measurement was plotted by using triplicate readings and the viability assay was plotted using duplicate readings. The data were analyzed for significance by unpaired Student’s t-test. *p<0.05, **p<0.001, ***p<0.0001, corresponding to the statistical difference between control and treated groups raw data were analyzed and plotted by Sigma Plot software.
Results
TB inhibits the Tau aggregation in vitro
The four repeat regions in Tau are considered aggregation-prone, VQIINK and VQIVYK hexapeptides present in the second and third repeat majorly contribute to the aggregation propensity of Tau. The repeat Tau has a basic charge with an isoelectric pH of 9.6 (Fig. 1A). TB is a basic dye belonging to the phenothiazine group of compounds (Fig. 1B). The aggregation inhibition property of TB was studied against heparin-induced Tau aggregation. The fluorescence studies suggested that TB efficiently inhibited Tau aggregation in a concentration-dependent manner. The inhibition was observed at a concentration of 2 µM and the rate of inhibition increased proportionally with the concentration of TB (Fig. 1C). 40 µM of TB was found to inhibit 80% of aggregation (p ≥0.001), suggesting TB is a potent molecule against Tau aggregation (Fig. 1D). The electron micrograph of TB-treated Tau showed broken and fragile fragment unlikely of the untreated sample, comprising of long and thick filamentous Tau aggregates (Fig. 1E-F).
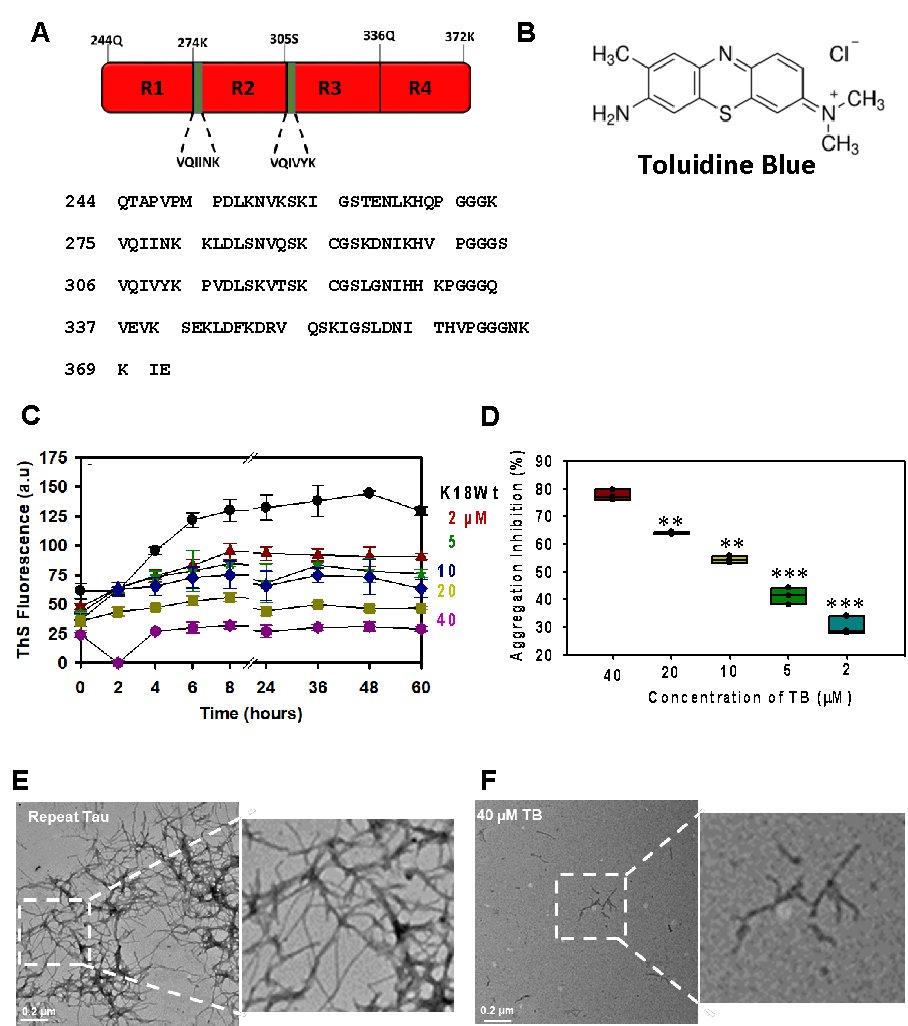
Fig. 1: The aggregation propensity of repeat Tau with Toluidine Blue. A) The four repeat region of Tau is prone to aggregation. TB is a phenothiazine dye having a basic charge. B) Aggregation of repeat Tau in presence of TB was probed by ThS fluorescence. The result suggested the aggregation inhibition of Tau in a concentration-dependent manner concerning TB. C-D) The endpoint fluorescence analysis to quantify the rate of aggregation inhibition by TB in a concentration-dependent manner. E, F) The electron microscopic analysis of aggregated Tau and TB-treated Tau. The significance was calculated using an unpaired Student’s t-test in SigmaPlot 10.0 software. *p < 0.05, **p < 0.001, ***p < 0.0001, the statistical difference between control and treated groups.
Photo-excited TB dissolved the matured Tau filaments
TB is a photo-sensitive dye having an absorption maxima of 630 nm. The irradiation of TB at 630±20 nm leads to the conversion of TB to photo-excited form, which generates singlet oxygen species (Fig. 2A). In our study, we exposed TB under a red LED light source for 180 minutes for photo-excitation with an irradiance of 9.9*106 Watt/m2. The Tau treated with PE-TB showed no higher-ordered aggregates bands on SDS-PAGE (Fig. 2B). The electron microscopy images supported the observation that PE-TB efficiently disaggregated the mature repeat Tau fibrils (Fig. 2C-D). Furthermore, ThS assay was carried out for evaluating the disaggregation potency of PE-TB. These results suggested that in comparison to untreated aggregates fluorescence intensity was decreased in PE-TB treated aggregates samples (Fig 2 E). In SDS-PAGE we did not observe any protein bands in PE-TB treated samples however, we observe decreased fluorescence in PE-TB treated samples which may be due to the small peptides which were not visible on SDS-PAGE. Additionally, in our experiments, negligible heat changes were observed after 180 minutes of irradiation. These in-vitro experiments indicated that PE-TB is an effective molecule against repeat Tau aggregates.
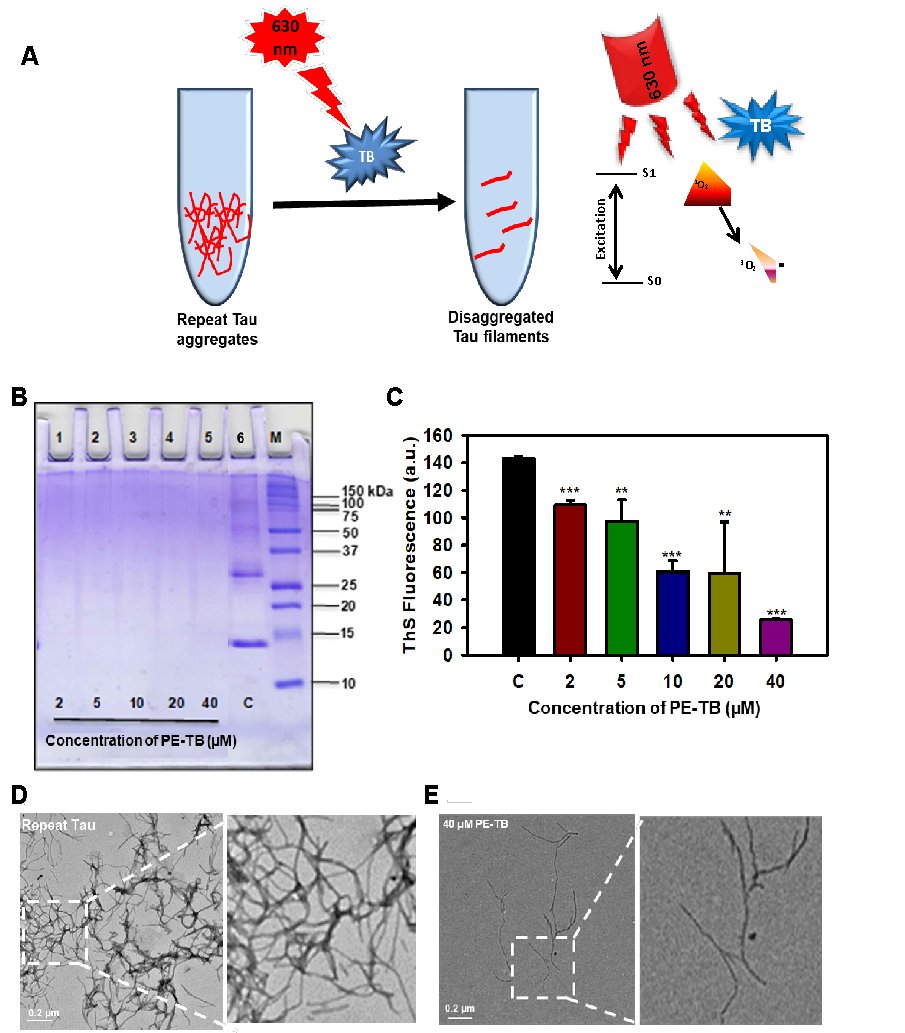
Fig. 2: Effect of PDT on repeat Tau aggregates at various concentrations of the photosensitizer. A) The repeat Tau aggregates were incubated with TB, which was further irradiated with 630 nm of red light. B) PE-TB (2-40 μM) on repeat Tau aggregates. The 17% SDS-PAGE shows the effect of PE-TB on higher molecular weight aggregates of repeat Tau. C) The ThS fluorescence shows the modulation in fluorescence intensity for PE-TB treated samples and untreated control. D, E) The electron microscopic images showing the morphologic changes in PE-TB treated aggregates vs. untreated control. The significance was calculated using Student’s t-test in SigmaPlot 10.0 software. *p < 0.05, **p < 0.001, ***p < 0.0001, the statistical difference between control and treated groups.
The cytotoxicity of higher concentrations of TB
The current experiment aimed to study the toxicity of TB and PE-TB against mouse neuroblastoma cells, Neuro2a. TB-treated cells were irradiated with 630±20 nm red light for 10 min with an irradiance of 5.5*105 Watt/m2 and incubated further for 24 hours. These results suggested that TB-induced low levels of toxicity than PE-TB. TB was found to have minimal toxicity at lower concertations, whereas a concentration higher than 20 µM induced significant toxicity. However, PE-TB was found to be toxic at a concentration of 20 µM (Fig. 3A-B). We speculated that the singlet oxygen produced by PE-TB might lead to the generation of toxicity. Furthermore at a concentration of 80 µM and above TB was observed to be internalized in neuronal cells (Fig. 3C). Here we speculate that TB was able to cross the cell membrane easily and thus the accumulation of higher concentrations of TB led to toxicity. The overall results suggested that TB has low toxicity even at higher concentrations.
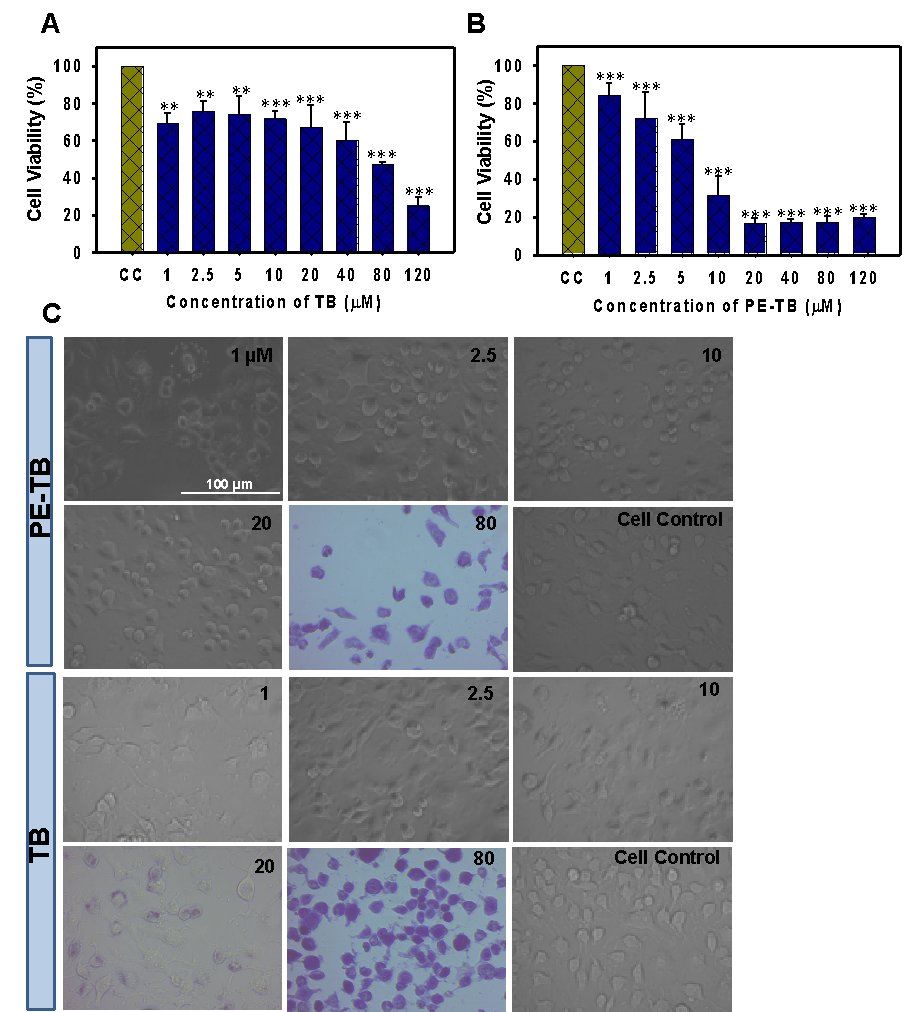
Fig. 3: The effect of a higher concentration of TB was observed on Neuro2A cell viability. A) The effect of non-photo-excited TB was observed on Neuro2A cells. B) Photo-excited TB was found to be toxic at higher concentrations. C) Morphological analysis of TB-treated cells. The significance was calculated using the Student’s t-test in SigmaPlot 10.0 software. *p < 0.05, **p < 0.001, ***p < 0.0001, the statistical difference between control and treated groups.
Modulation of the cytoskeleton by PE-TB
Tubulin is a basic unit of the cytoskeleton, which polymerizes leading to the formation of microtubules. The effect of PE-TB on the cytoskeleton was observed by immunofluorescence studies. Neuro2a cells were incubated for 24 hours with varying concentrations of PE-TB (0.5 and 50 µM). The immunofluorescence studies suggested that as compared to untreated cell control, cells treated with 0.5 µM PE-TB have high fluorescence intensity of tubulin, whereas no differential results were observed in cells treated with 50 µM of PE-TB treatment (Fig. 4A-B). The quantification of fluorescence images suggested a minimal increase in tubulin intensity. Tubulin is the basic unit of microtubules thus, the modulation of tubulin intensity suggested modification of the cytoskeleton after PE-TB treatment (Fig. 4C). Although the morphological changes in the cell after PE-TB treatment was not appreciably significant minimal changes in elongated dendritic extensions were observed in treated cells (Fig. 4D). Moreover, to analyze the effect of TB and PE-TB on cytoskeleton we analyzed the actin modulation (Fig. 5A). Our studies indicated that TB treatment induces actin structure in cells, as the cells were observed to have actin-rich “Lamellipodia” structures after TB exposure. While the cells treated with PE-TB were observed to have increased lamellipodia structures, which supported the actin modulation (Fig. 5B). Furthermore, we studied the End-binding protein 1 (EB1) expression in neurons. These results suggested that TB and PE-TB treatment enhanced the levels of EB1, which suggested that TB and PE-TB treatment might accelerate the microtubule polymerization in cells (Fig. 6A-B). The elevated levels of EB1 indicated that there could be an increased rate of microtubule polymerization after the TB and PE-TB treatment.
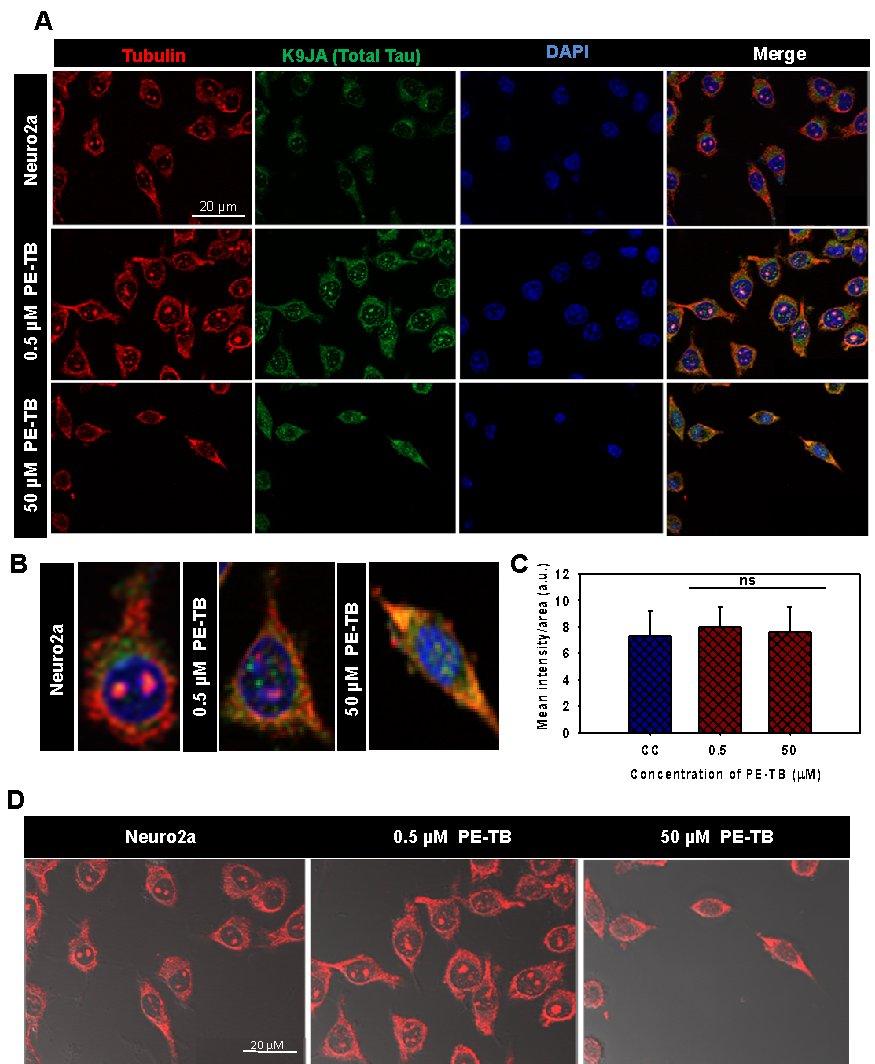
Fig. 4: The effect of PE-TB on the cytoskeleton. A) The effect of PE-TB on the cytoskeleton was studied by immunofluorescence. The cells treated with various contritions of PE-TB (0.5 µM and 50 µM) showed modulation in tubulin intensity. B) The single-cell images showing the differential level of tubulin in PE-TB treated cells. C) The quantification of immunofluorescence images suggested that the intensity of tubulin increased after PE-TB treatment to cells. D) Images including DIC showing the morphological changes after PE-TB treatment of Neuro2A cells.
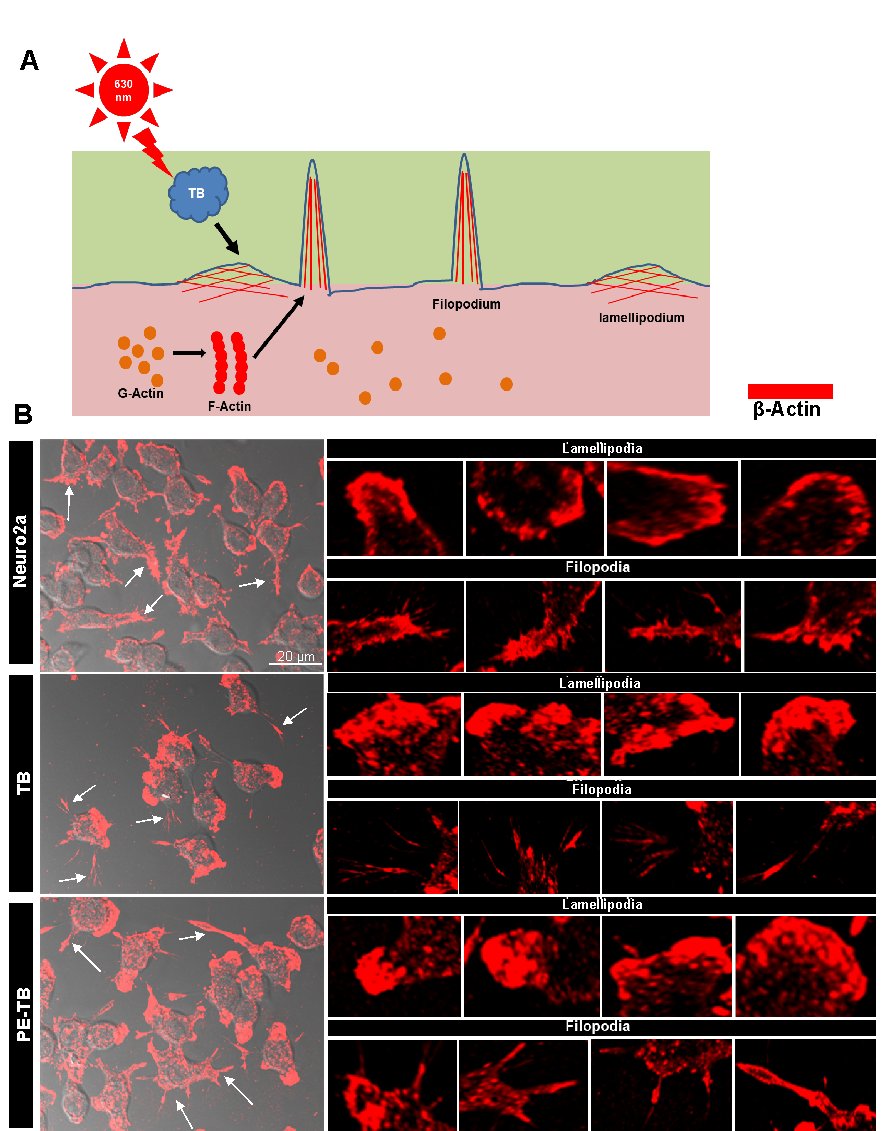
Fig. 5: TB induces modulation in the cytoskeleton. A) The schematic diagram for the alteration of actin cytoskeleton after TB and PE-TB treatment. B) The immunofluorescence images showing the changes in the actin cytoskeleton, the filopodia (arrow marked), and lamellipodia (star marked) structures after TB and PE-TB treatment.
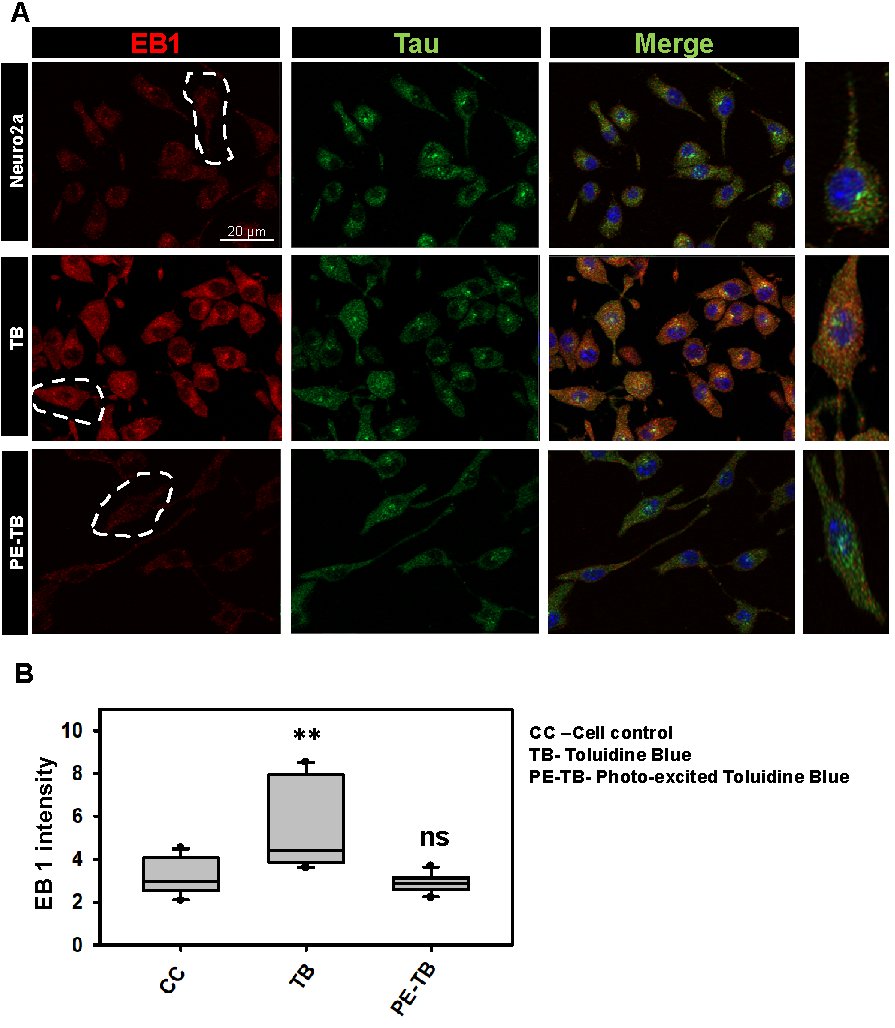
Fig. 6: TB and PE-TB induce changes in EB1 expression. A) The immunofluorescence images show the changes in EB1 levels after TB treatment. B) The quantification of immunofluorescence images suggests that TB increases the EB1 levels in cells. The quantification was done by using Zen 2.0 blue software.
Discussion
The pathological state of Tau leads to the formation of Tau aggregates, several factors including post-translational modifications, reactive oxygen species, and mutations result in the generation of Tauopathies [3, 26]. Several studies have been done on the screening of the compounds against Tauopathy [27]. Various classes of dyes were tested for their potency against neurodegeneration. The aggregation inhibition potency of methylene blue for Tau has been already reported illustratively. Xanthene dye such as erythrosine B was reported to reduce the Amyloid-β-mediated toxicity in cells. Photo-excited rose bengal was found potent in inhibiting the Amyloid-β aggregation in liver cells. Furthermore, the sulfonated dye congo red was reported to attenuate the Amyloid-β aggregation. The phenothiazine class of dyes was reported to have therapeutic potency against numerous diseases [28]. Here we have studied the efficiency of TB against Tau aggregation and our observations suggested that as its parent compound methylene blue, TB was also efficient in inhibiting the aggregation of repeat Tau. Recently researchers have investigated that irradiation plays a crucial role in the treatment of AD. Moreover, EGCG irradiated with near-infra-red light was found to reduce the Amyloid-β plaque deposition in neuronal cells [29]. In our studies, we irradiated TB with 630 nm red light to study its effect on Tau aggregates. Earlier studies have reported that TB reduces Amyloid-β aggregation but the effect of TB was found ineffective against Tau hyperphosphorylation [30]. We found that PE-TB potentially disaggregated the pre-formed Tau filaments. The PE-TB led to the generation of singlet oxygen species, thus we speculate that singlet oxygen contributes to the disaggregation potency of TB. The SDS-PAGE, ThS analysis, and electron microscopic studies indicated the disaggregation of Tau filaments by PE-TB. The phenothiazine dye-induced low levels of toxicity in cells, but on photo-irradiation, the dyes induced toxicity in cells due to the generation of singlet oxygen species [31]. In our work, we found that TB was not significantly toxic to cells even at high concentrations of 20 µM but the PE-TB reduced the viability of cells at concertation of 20 µM. These results suggested that TB at lower concentrations could be considered biocompatible. The potency of photo-irradiation in modulating the cytoskeleton has been reported in human hepatoma cells [32]. The reports suggested that irradiation inhibited cell growth by modulating the cytoskeleton. In our results, we observed that PE-TB treatment minimally increases the tubulin intensity in neuronal cells, which indicated that PE-TB could modulate the cytoskeleton structure. The actin cytoskeleton plays a crucial role in cell motility and cell synapse formation [33]. The lamellipodia are the actin-rich structures and the modulation in lamellipodia led to changes in cell motility and adhesion [9]. Similarly, the modulation in filopodia structure alters the cell synapse formation and motility [34]. Thus, the alterations in the actin network after TB and PE-TB treatment led us to the conclusion that the dye has the potency to modulate cell cytoskeleton networks. Hence, the overall results suggest that PE-TB could have a therapeutic potency against Tauopathy (Fig. 7).
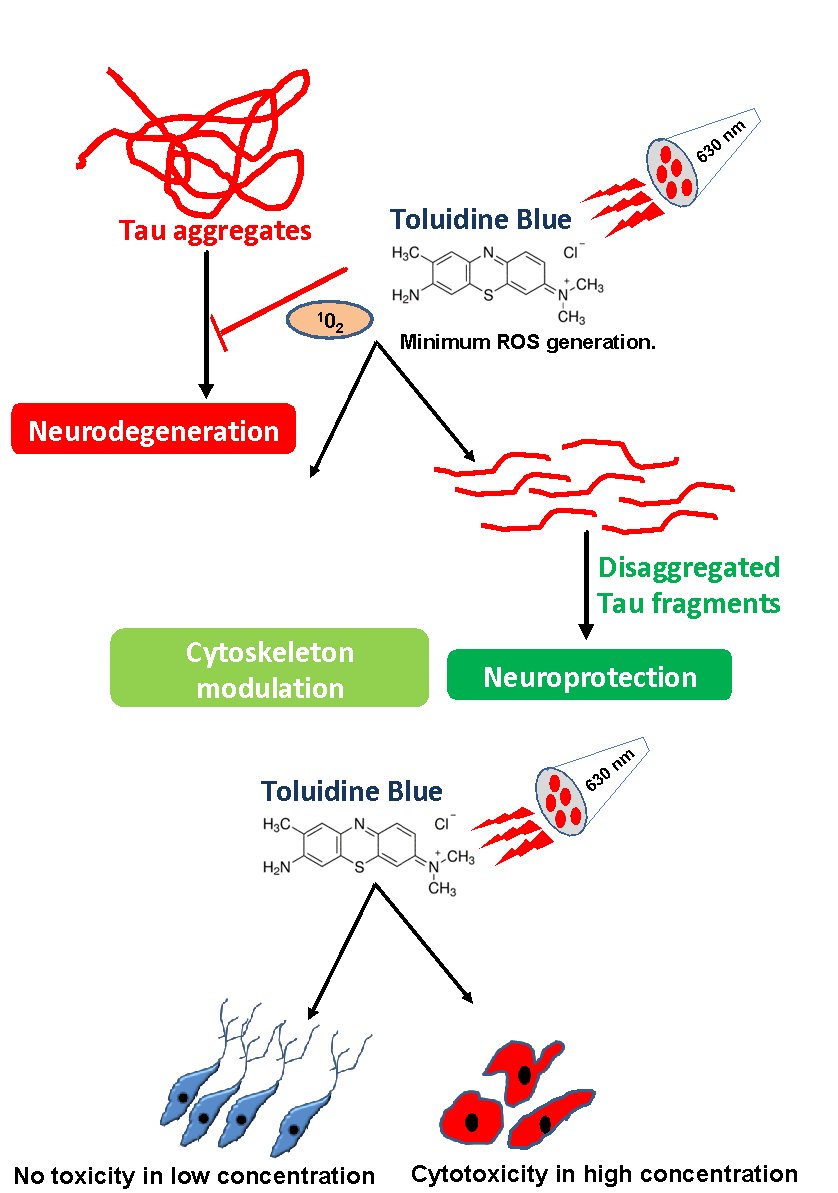
Fig. 7: The TB and PE-TB dissolve the Tau fibrils. Tau aggregates are one of the hallmarks of AD. Accumulation of Tau aggregates leads to the generation of neurodegenerative diseases. TB on irradiation with 630 nm red light gets converted to PE-TB. PE-TB disaggregates the pre-formed Tau and modulates the cytoskeleton. Thus, PE-TB could be a potent molecule against Tauopathy. The cytotoxicity studies suggest that in lower concentrations TB and PE-TB have no adverse effect on cell viability.
Abbrevations
AD (Alzheimer’s disease); BCA (Bicinchoninic acid); BSA (Bovine serum albumin); DMSO (Dimethyl sulfoxide); MB (Methylene blue); MTT (Methylthiazolyldiphenyl-tetrazolium bromide); NFTs (Neurofibrillary tangles); PHFs (Paired helical filaments); PDT (Photodynamic therapy); PE-TB (Photo-excited TB); SEC (Size exclusion chromatography); SDS-PAGE (Sodium dodecyl sulfate-polyacrylamide gel electrophoresis); TEM (Transmission electron microscopy); TB (Toluidine blue); ThS (Thioflavin S).
Acknowledgements
The authors acknowledge CSIR-National Chemical Laboratory for the instrumentation central facility. Tushar Dubey acknowledges the fellowship from University of Grant Commission (UGC), India. Authors greatly acknowledged Nalini Vijay Gorantla for her initial experiements on Tau and RB.
Ethical approval
Not applicable. There were no animal experiments have been carried out in the current manuscript.
Consent for publication
Not applicable.
Availability of data and material
All the data generated during various experiments are available from the authors upon reasonable request.
Data Availability
The data that support the findings of this study are available in this manuscript.
Funding
This project is supported by grant from the in-house, National Chemical Laboratory-Council of Scientific Industrial Research CSIR-NCL MLP029526.
Author Contributions
SBC designed the experiments. TD and SBC carried out the experiments. TD and SBC analyzed the data and wrote the manuscript. M.C helped in the analysis and revision of the manuscript. SBC conceived the idea of the project, provided resources, supervised and wrote the manuscript. All authors contributed to the discussions and manuscript review.
Author Information
Subashchandrabose Chinnathambi and Tushar Dubey
Neurobiology Group, Division of Biochemical Sciences, CSIR-National Chemical Laboratory, Dr. Homi Bhabha Road, Pune, 411008, India
Tushar Dubey, and Subashchandrabose Chinnathambi
Academy of Scientific and Innovative Research (AcSIR), Ghaizabad, 201002, India
Department of Neurochemistry, National Institute of Mental Health and Neuro Sciences (NIMHANS), Institute of National Importance, Hosur Road, Bangalore -560029, Karnataka, India.
Madhura chnadrashekar
The School of Bioengineering Sciences and Research, MIT Art, Design & Technology, University, Loni Kalbhor, 412201, Pune, India
Disclosure Statement
The authors declare that they have no competing interest.
References
| 1 | Mandelkow E-M, Mandelkow E: Biochemistry and cell biology of tau protein in neurofibrillary degeneration. Cold Spring Harb Perspect Med 2012:a006247.
https://doi.org/10.1101/cshperspect.a006247 |
| 2 | Gorantla NV, Shkumatov AV, Chinnathambi S: Conformational dynamics of intracellular tau protein revealed by CD and SAXS. Tau Protein. Methods Mol Biol 2017;1523:3-20.
https://doi.org/10.1007/978-1-4939-6598-4_1 |
| 3 | Iqbal K, Alonso AdC, Chen S, Chohan MO, El-Akkad E, Gong C-X, Khatoon S, Li B, Liu F, Rahman A: Tau pathology in Alzheimer disease and other tauopathies. Biochim Biophys Acta Mol Basis Dis 2005;1739:198-210.
https://doi.org/10.1016/j.bbadis.2004.09.008 |
| 4 | Sonawane SK, Chinnathambi S: Prion-Like Propagation of Post-Translationally Modified Tau in Alzheimer’s Disease: A Hypothesis. J Mol Neurosci 2018;65:480-490.
https://doi.org/10.1007/s12031-018-1111-5 |
| 5 | Mitchison T, Cramer L: Actin-based cell motility and cell locomotion. Cell 1996;84:371-379.
https://doi.org/10.1016/S0092-8674(00)81281-7 |
| 6 | Bamburg JR, Bloom GS: Cytoskeletal pathologies of Alzheimer disease. Cell motil cytoskelet 2009;66:635.
https://doi.org/10.1002/cm.20388 |
| 7 | Cairns NJ, Lee VMY, Trojanowski JQ: The cytoskeleton in neurodegenerative diseases. The Journal of Pathology: A Journal of the Pathological Society of Great Britain and Ireland 2004;204:438-449.
https://doi.org/10.1002/path.1650 |
| 8 | Hadfield JA, Ducki S, Hirst N, McGown AT: Tubulin and microtubules as targets for anticancer drugs. Prog Cell Cycle Res 2003;5:309-326.
|
| 9 | Small JV, Stradal T, Vignal E, Rottner K: The lamellipodium: where motility begins. Trends Cell Biol 2002;12:112-120.
https://doi.org/10.1016/S0962-8924(01)02237-1 |
| 10 | Stepanova T, Slemmer J, Hoogenraad CC, Lansbergen G, Dortland B, De Zeeuw CI, Grosveld F, van Cappellen G, Akhmanova A, Galjart N: Visualization of microtubule growth in cultured neurons via the use of EB3-GFP (end-binding protein 3-green fluorescent protein). J Neurosci 2003;23:2655-2664.
https://doi.org/10.1523/JNEUROSCI.23-07-02655.2003 |
| 11 | Ballatore C, Lee VM-Y, Trojanowski JQ: Tau-mediated neurodegeneration in Alzheimer’s disease and related disorders. Nat Rev Neurosci 2007;8:663.
https://doi.org/10.1038/nrn2194 |
| 12 | Balmik AA, Chinnathambi S: Multi-faceted role of melatonin in neuroprotection and amelioration of Tau aggregates in Alzheimer’s disease. J Alzheimers Dis 2018;62:1481-1493.
https://doi.org/10.3233/JAD-170900 |
| 13 | Gorantla NV, Landge VG, Nagaraju PG, Priyadarshini CG P, Balaraman E, Chinnathambi S: Molecular cobalt (II) complexes for tau polymerization in Alzheimer’s disease. ACS omega 2019;4:16702-16714.
https://doi.org/10.1021/acsomega.9b00692 |
| 14 | Khlistunova I, Biernat J, Wang Y, Pickhardt M, von Bergen M, Gazova Z, Mandelkow E, Mandelkow E-M: Inducible expression of Tau repeat domain in cell models of tauopathy aggregation is toxic to cells but can be reversed by inhibitor drugs. J Biol Chem 2006;281:1205-1214.
https://doi.org/10.1074/jbc.M507753200 |
| 15 | Gorantla NV, Das R, Mulani FA, Thulasiram HV, Chinnathambi S: Neem Derivatives Inhibits Tau Aggregation. J Alzheimers Dis Rep 2019;3:169-178.
https://doi.org/10.3233/ADR-190118 |
| 16 | Pickhardt M, Gazova Z, von Bergen M, Khlistunova I, Wang Y, Hascher A, Mandelkow E-M, Biernat J, Mandelkow E: Anthraquinones inhibit tau aggregation and dissolve Alzheimer’s paired helical filaments in vitro and in cells. J Biol Chem 2005;280:3628-3635.
https://doi.org/10.1074/jbc.M410984200 |
| 17 | Sonawane SK, Balmik AA, Boral D, Ramasamy S, Chinnathambi S: Baicalein suppresses Repeat Tau fibrillization by sequestering oligomers. Arch Biochem Biophys 2019;675:108119.
https://doi.org/10.1016/j.abb.2019.108119 |
| 18 | Lee BI, Suh YS, Chung YJ, Yu K, Park CB: Shedding light on Alzheimer’s β-amyloidosis: photosensitized methylene blue inhibits self-assembly of β-amyloid peptides and disintegrates their aggregates. Sci Rep 2017;7:7523.
https://doi.org/10.1038/s41598-017-07581-2 |
| 19 | Lee JS, Lee BI, Park CB: Photo-induced inhibition of Alzheimer’s β-amyloid aggregation in vitro by rose bengal. Biomater 2015;38:43-49.
https://doi.org/10.1016/j.biomaterials.2014.10.058 |
| 20 | Kostelanska M, Freisleben J, Backovska Hanusova Z, Mosko T, Vik R, Moravcova D, Hamacek A, Mosinger J, Holada K: Optimization of the photodynamic inactivation of prions by a phthalocyanine photosensitizer: the crucial involvement of singlet oxygen. J Biophotonics 2019:e201800340.
https://doi.org/10.1002/jbio.201800430 |
| 21 | Leshem G, Richman M, Lisniansky E, Antman-Passig M, Habashi M, Gräslund A, Wärmländer SK, Rahimipour S: Photoactive chlorin e6 is a multifunctional modulator of amyloid-β aggregation and toxicity via specific interactions with its histidine residues. Chem 2019;10:208-217.
https://doi.org/10.1039/C8SC01992D |
| 22 | Wilcock GK, Gauthier S, Frisoni GB, Jia J, Hardlund JH, Moebius HJ, Bentham P, Kook KA, Schelter BO, Wischik DJ: Potential of low dose leuco-methylthioninium bis (hydromethanesulphonate)(LMTM) monotherapy for treatment of mild Alzheimer’s disease: Cohort analysis as modified primary outcome in a phase III clinical trial. Journal of Alzheimer’s Disease 2018;61:435-457.
https://doi.org/10.3233/JAD-170560 |
| 23 | Sun B-L, Li W-W, Zhu C, Jin W-S, Zeng F, Liu Y-H, Bu X-L, Zhu J, Yao X-Q, Wang Y-J: Clinical research on Alzheimer’s disease: progress and perspectives. Neuroscience bulletin 2018;34(6):1111-18.
https://doi.org/10.1007/s12264-018-0249-z |
| 24 | Biberoglu K, Tek MY, Ghasemi ST, Tacal O: Toluidine blue O is a potent inhibitor of human cholinesterases. Arch Biochem Biophys 2016;604:57-62.
https://doi.org/10.1016/j.abb.2016.06.005 |
| 25 | Dubey T, Gorantla NV, Chandrashekara KT, Chinnathambi S: Photoexcited toluidine blue inhibits Tau aggregation in Alzheimer’s disease. ACS omega 2019;4:18793-18802.
https://doi.org/10.1021/acsomega.9b02792 |
| 26 | Iqbal K, Liu F, Gong C-X: Tau and neurodegenerative disease: the story so far. Nat Rev Neurol 2016;12:15-27.
https://doi.org/10.1038/nrneurol.2015.225 |
| 27 | Gorantla NV, Landge VG, Nagaraju PG, Sunny LP, Nair A, Midya SP, Priyadarshini P, Ekambaram B, Chinnathambi S: Molecular Complexes for Effective Inhibition of Tau Aggregation. bioRxiv 2018:363572.
https://doi.org/10.1101/363572 |
| 28 | PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Patent Summary for CA-2827027-C, Phenothiazine diaminium salts and their use; [cited 2023 Apr. 14]. Available from: https://pubchem.ncbi.nlm.nih.gov/patent/CA-2827027-C
|
| 29 | Johnstone DM, Moro C, Stone J, Benabid A-L, Mitrofanis J: Turning on lights to stop neurodegeneration: the potential of near infrared light therapy in Alzheimer’s and Parkinson’s disease. Front Neurosci 2016;9:500.
https://doi.org/10.3389/fnins.2015.00500 |
| 30 | Yuksel M, Biberoglu K, Onder S, Akbulut KG, Tacal O: Toluidine blue O modifies hippocampal amyloid pathology in a transgenic mouse model of Alzheimer’s disease. Biochimie 2018;146:105-112.
https://doi.org/10.1016/j.biochi.2017.12.004 |
| 31 | Wainwright M, Phoenix DA, Rice L, Burrow SM, Waring J: Increased cytotoxicity and phototoxicity in the methylene blue series via chromophore methylation. J Photochem Photobiol B 1997;40:233-239.
https://doi.org/10.1016/S1011-1344(97)00061-4 |
| 32 | Liu Y-H, Ho C-C, Cheng C-C, Hsu Y-H, Lai Y-S: Photoradiation could influence the cytoskeleton organization and inhibit the survival of human hepatoma cells in vitro . Lasers in medical science 2006;21(1):42.
https://doi.org/10.1007/s10103-005-0369-3 |
| 33 | Luo L: Actin cytoskeleton regulation in neuronal morphogenesis and structural plasticity. Annu Rev Cell Dev Biol 2002;18:601-635.
https://doi.org/10.1146/annurev.cellbio.18.031802.150501 |
| 34 | Mallavarapu A, Mitchison T: Regulated actin cytoskeleton assembly at filopodium tips controls their extension and retraction. J Cell Biol 1999;146:1097-1106.
https://doi.org/10.1083/jcb.146.5.1097 |
| 35 | Gorantla NV, Balaraman E, Chinnathambi S: Cobalt-based metal complexes prevent Repeat Tau aggregation and nontoxic to neuronal cells. Int J Biol Macromol 2020;152:171-179.
https://doi.org/10.1016/j.ijbiomac.2020.02.278 |
| 36 | Gorantla NV, Das R, Balaraman E, Chinnathambi S: Transition metal nickel prevents Tau aggregation in Alzheimer’s disease. Int J Biol Macromol 2020;156:1359-1365.
https://doi.org/10.1016/j.ijbiomac.2019.11.176 |
| 37 | Sonawane SK, Ahmad A, Chinnathambi S: Protein-Capped Metal Nanoparticles Inhibit Tau Aggregation in Alzheimer’s Disease. ACS Omega 2019;4:12833-12840.
https://doi.org/10.1021/acsomega.9b01411 |
| 38 | Gorantla NV, Chinnathambi S: Tau protein squired by molecular chaperones during Alzheimer’s disease. J Mol Neurosci 2018;66:356-368.
https://doi.org/10.1007/s12031-018-1174-3 |